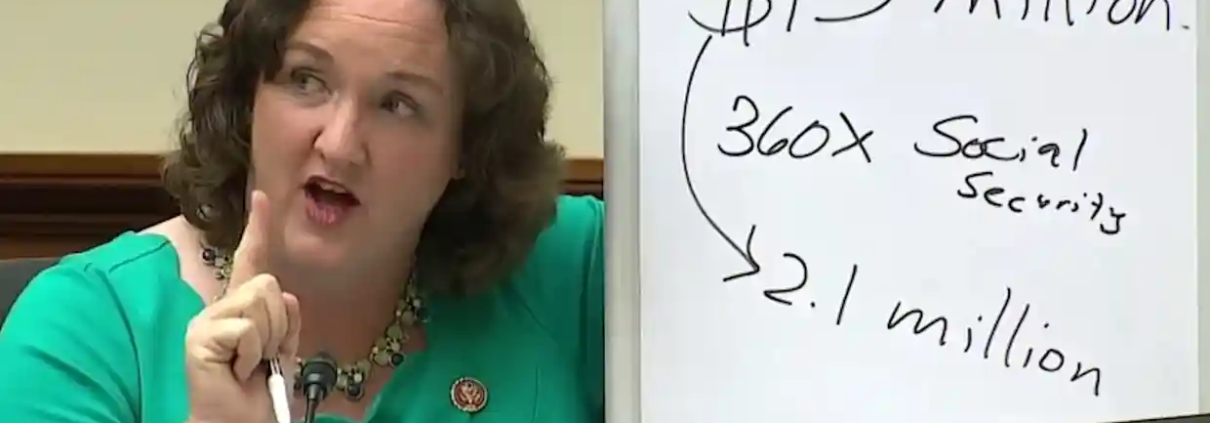Pharma’s Post-Pandemic Policy Outlook
Here’s What You Need to Know
As the first COVID-19 vaccines were delivered this week, pharmaceutical industry leaders recognize that there are still many challenges ahead. While the industry overcame the herculean task of developing and distributing a vaccine in unprecedented time, the pandemic has brought previously obscure issues such as international patent rights and the global supply chain of pharmaceutical ingredients to the forefront of major public policy debates worldwide. With this heightened scrutiny and increased pressure from policymakers and the public, the pharmaceutical industry will have to continue to navigate these debates in 2021 and beyond.
To help prepare pharmaceutical industry public affairs professionals for what 2021 will bring, we will be launching a new occasional trends report in January focused on the political and reputational risks facing the industry. Below is a preview of some of the issues the trends report will cover, and we hope you will sign up to receive full editions of the report when they are published.
Cross-Partisan Pressure and Scrutiny on Drug Prices Will Continue in the Biden Era
Prior to the pandemic, policy makers on both sides of the isle debated how to address what many perceive as the high price of pharmaceuticals. While in most areas the Biden administration is likely to divert from President Trump’s policy direction, in this debate there may be more continuity than conflict. In September of this year President Trump signed an . While this will likely end up being challenged in court, it likely will not deter the incoming Biden administration from seeking to advance a similar approach to reduce drug prices, which was also included in the House Democrats’ drug pricing plan endorsed by progressive groups.
Subscribe to Receive Insights
"*" indicates required fields
Beyond this specific proposal, pharmaceutical companies can expect more scrutiny from Washington. President-elect Joe Biden’s pick to lead Health and Human Services (HHS), California Attorney General In addition, Congress is likely to continue oversight hearings similar to those seen earlier this year and in prior years seeking to question the industry’s practices and pricing with
The Pandemic Has Globalized the Fight Over Patent Rights as Activists Seek to Upend the Industry
As the industry’s search for COVID-19 cures and treatments began in earnest, a People’s Vaccine movement emerged to demand any vaccine be made a “global public good” available for free and equitably distributed to both the developing and developed world simultaneously. This movement didn’t crop up overnight. For years, activists and groups advancing this campaign have been involved in fights across the developing world advocating for compulsory licensing laws to increase access to medicines. The People’s Vaccine movement has culminated with a fierce campaign advocating for World Trade Organization member states to back a proposal by India and South Africa for a waiver on intellectual property related to COVID-19 and calling for wealthy countries and pharmaceutical companies to back international intellectual property pools. This proposal remains under consideration and the debate over it will continue into 2021.
This movement to challenge patent rights has been building for a long time in the developing world, typically focused on specific regions and access to drugs that treat HIV/AIDS, Hepatitis, Tuberculosis, and other illnesses where advances in medical technology have not always been widely available in low and middle income countries. However, COVID has allowed this movement to become more global in its focus as it seeks to leverage the pandemic to reshape the model of how we fund and commercialize health technology. As one prominent activist in that movement, Medicines Law & Policy Director Ellen ‘t Hoen, recently stated, “We should not lose sight of the need to work to increase access to other new essential medicines. … After COVID, I believe cancer will prove to be the next big challenge,” with treatments “priced well beyond the ability to pay for most people and communities in the developing world.”
2020 Brought Heightened Scrutiny on Drug Supply Chains. 2021 Could Bring Action To Force Them Home.
As we wrote this past spring, the pandemic “exposed cracks in the worldwide supply chain for pharmaceuticals and medical supplies necessary to create and administer vaccines.” In hopes of addressing this, President-elect Biden has made clear that one of the goals of his administration will be to bring pharmaceutical supply chains and manufacturing back to the United States. On the campaign trail, Biden endorsed the creation of a national “Critical Drugs List” as part of an overarching plan to address supply chains shortages and encourage US-based manufacturing of prescription drugs.
Meanwhile, Congressional Republicans have joined bipartisan calls to spur greater U.S. drug manufacturing. This shift in approach by many free market-oriented Republicans stems from growing voter concerns regarding globalization as well as the national security threats associated with reliance on China – both of which have been exacerbated by COVID-19. However, the price tag that comes attached to bringing these supply chains and manufacturing abilities back to the United States has some industry leaders concerned, with one leader saying that it is “unrealistic” to think that it could be done in part to due to steep regulatory hurdles, while others
Stay Ahead With Actionable Insights
As 2020 comes to a close and we approach the inauguration of new administration, the challenges the pharmaceutical industry will face in the year(s) ahead have come to the forefront of the public policy debate. Our team of analysts will continue to dig deeper and monitor developments that will impact the industry. Sign up for our trends report to keep you ahead of the curve, and feel free to reach out for more in-depth analysis on any or all of these challenges and opportunities facing the industry.